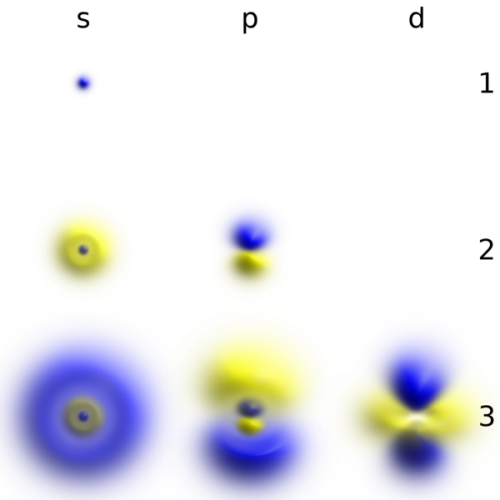
10 Facts about Electrons
Facts about Electrons define the subatomic particle with negative electric charge. The symbol of electron in e-. The researchers find out that electrons do not have any substructures or components. Therefore, they are included as elementary particles. In the lepton particle family, it belongs to the first generation. The mass of an electron is smaller compared to proton. It only has the mass of 1/1836 than the mass of a proton. A case where two electrons will occupy the similar quantum state is not possible because electron is a fermion. Find out other interesting facts about electrons below:
Facts about Electrons 1: the properties
The waves and particles are the two common properties that electrons have. Electrons the same nature like light which can be diffracted. You can also spot electrons colliding with other particles.
Facts about Electrons 2: observation
Observing the wave electron is much easier than the proton and neutron due to the light mass that it has.
Facts about Electrons 3: the importance of electrons
Electron is important in the world of science since this term means anything in various fields of studies such as thermal conductivity, chemistry, magnetism, electricity. During the electromagnetic and gravitational interactions, the electrons play an important role.
See Also: 10 Facts about Electrical Safety
Facts about Electrons 4: electric field
The electric field is found at the surrounding area of an electron as it has charge. A magnetic field is produced when the electron is moving relative to a viewer.
Facts about Electrons 5: the Lorentz force law
Have you ever heard about the Lorentz force law? The law states that the motion of an electron will be affected by the electromagnetic field generated from other sources.
Facts about Electrons 6: acceleration
Acceleration will affect the electrons. They will absorb or radiate the energy in the form of photons. By using electromagnetic, the scientists can trap the electron plasma and electrons individually in the laboratory.
Facts about Electrons 7: the application of electrons
Electrons have various applications in science. The particle accelerators, gaseous ionization detectors, radiation therapy, cathode ray tube, welding, electronics and electron microscopes are involved with the use electrons. When a scientist wants to check the electron plastic in the space, they can employ a special telescope.
Related Article: 10 Facts about Electrical Engineering
Facts about Electrons 8: the study of electrons
Chemistry and nuclear physics are the two primary fields of study where the students can learn more about the interaction between electrons and subatomic particles.
Facts about Electrons 9: the chemical bonding
The chemical bonding occurs because two or more atoms share or exchange electrons. In 1891, the term electron was coined by George Johnstone Stoney. He was a physicist from Ireland. In 1897, it was defined as a particle by J.J. Thomson and his team.
Facts about Electrons 10: experiments
The physicists like Ernest Rutherford, Gustav Hertz, James Franck and Henry Moseley made experiments in 1914, which found out that the lower mass electrons were around a dense nucleus of positive charge.
Are you well informed after reading facts about electrons?